Better health through innovation!

Better health through innovation!


OUR PROJECTS
2013 - Use of Various TLR Agonists as Adjuvants in improving Food & Mouth Disease Efficacy, Supported by the Ministry of Science, Industry and Technology, Techno-initiative Fund.
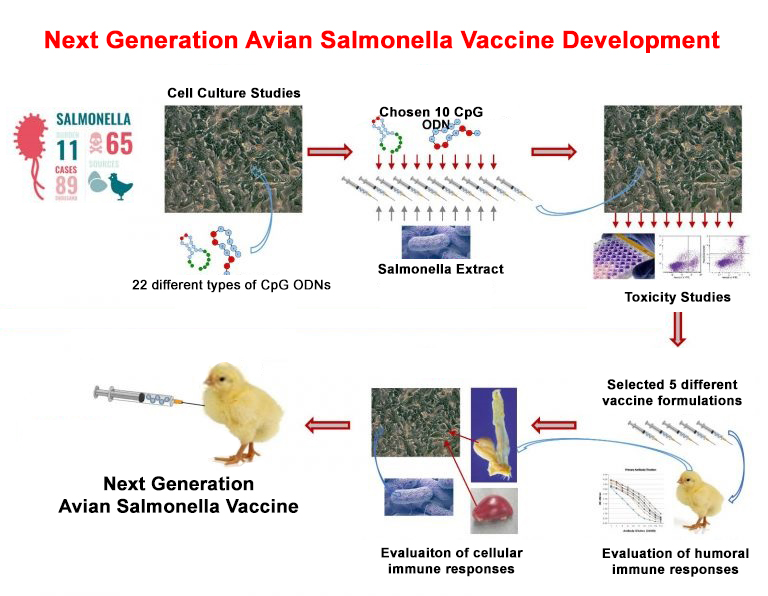
2014 - Use of CpG ODN’s as adjuvants in Avian Salmonella vaccine formulations. Supported by the Ministry of Science, Industry and Technology, Techno-initiative Fund.
2014 - Development of adjuvants for use in increasing vaccine efficacy.
2014 - Development of prostate cancer detection kits using extracellular vesicles in urine. Funded by the 1512 Entrepreneurial Support Program.
2017 - Nucleic acid based protective and therapeutic Hepatits B vaccine.
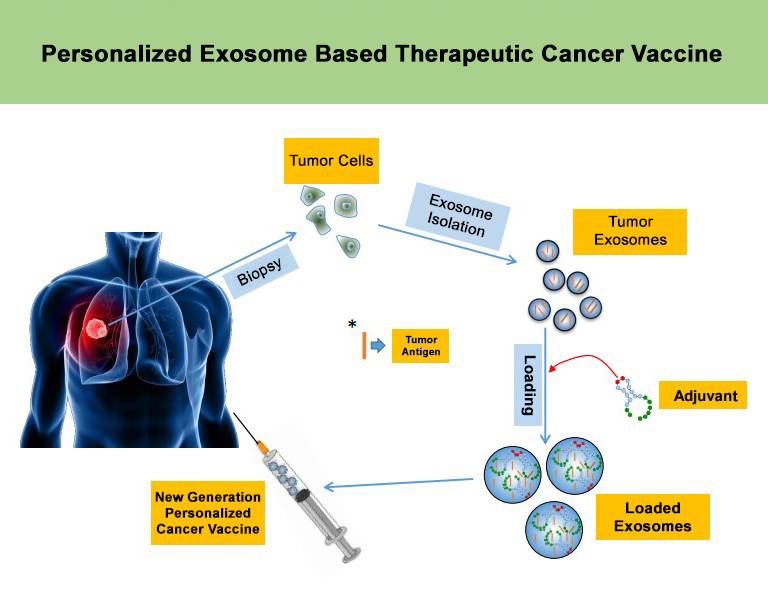
2019 - Personalized exosome based therapeutic cancer vaccine.
2021 - Development of ELISA & Flow Cytometry Diagnostic Kits for COVID-19. Funded by the Turkish FDA 1501 Coded Industry R&D Support Program.
2023 - Development of Cytokine and Kemokine pre-coated ELISA plates for Research.
1. Yazar V, Kilic G, Bulut O, Yildirim Canavar T, Yagci FC, Aykut G, Klinman DM, Gursel M, Gursel I. “A suppressive oligodeoxynucleotide expressing TTAGGG motifs modulates cellular energetics through the mTOR signaling pathway”. Int Immunol. October 2019.
2. Kahraman T, Gucluler G, Simsek I, Yagci FC, Yildirim M, Ozen C, Dinc A, Gursel M, Ikromzoda L, Sutlu T, Gay S, Gursel I. “ Circulating LL37 targets plasma extracellular vesicles to immune cells and intensifies Behcet’s disease severity”. J Extracell Vesicles 28;6(1), February 2017.
3. Gungor B, Yagci FC, Gursel I, Gursel M. “Forging of a Potent Vaccine Adjuvant: CpG ODN/Cationic Peptide Nanorings”. Oncoimmunology, 3;3(7), July 2014.
4. Gungor B*, Yagci FC*, Tincer G, Bayyurt B, Alpdundar E, Yildiz S, Ozcan M, Gursel I, Gursel M. “CpG ODN nanorings induce IFNα from plasmocytoid dendritic cells and demonstrate potent vaccine adjuvant activity”. Sci Transl Med. 7;6 (235):235ra61, May 2014.
* These authors contributed equally to this work.
5. Osmanagaoglu O, Kıran F, Yagci FC, Gursel I. “Immunomodulatory function and in vivo properties of Pediococcus pentosaceus OZF, a promising probiotic strain.” Ann Microbiol DOI 10.1007/s13213-012-0590-9, December 2012.
6. Tincer G, Yerlikaya S, Yagci FC, Kahraman T, Atanur OM, Erbatur O, Gursel I. “Immunostimulatory Activity of Polysaccharide-Poly(I:C) Nanoparticles.” Biomaterials. 2011 Jun;32(18):4275-82, June 2011.
7. Yagci FC, Aslan O, Gursel M, Tincer G, Ozdamar Y, Karatepe K, Akcali CK, Gursel I. “Mammalian Telomeric DNA Suppresses Endotoxin Induced Uveitis”. J Biol Chem. 10;285(37):28806-11, September 2010.

M E N U